FDA decision could come within weeks, but big obstacles remain to getting all children inoculated.
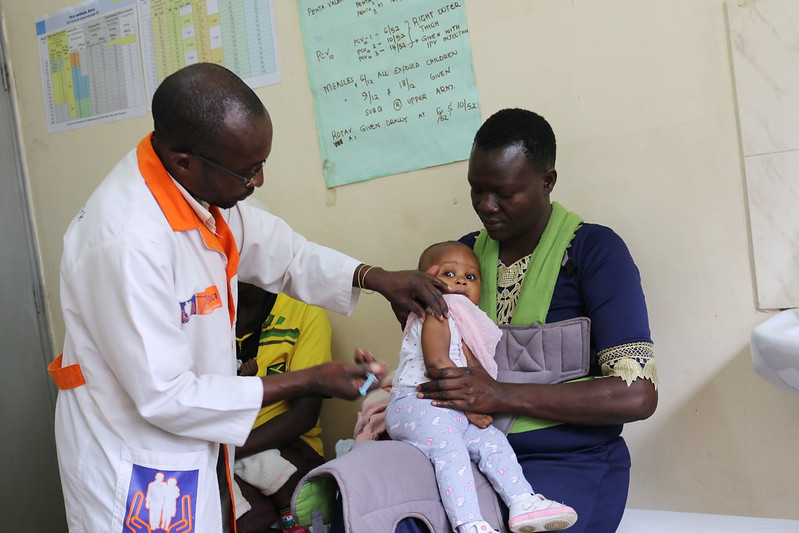
Children under five, the last group of Americans still ineligible for vaccines against Covid-19, may soon receive emergency authorization for the shots, but getting all children vaccinated remains a serious challenge in the US.
Pfizer and its German pharmaceutical partner BioNTech announced on Tuesday that they were requesting emergency-use authorization of their vaccine for children aged six months to four years.
The application was submitted at the request of the US Food and Drug Administration, the companies said in a statement – an unusual move by the regulator. Read more at the Guardian.
Categories: Freelance Articles







